Will ammonia kill aquarium plants? This question bother many aquarists for years. I am going to explain the answer in this article. In the end, you will know more about ammonium in fish tanks.

Will ammonia kill aquarium plants?
Jump To
Yes, excess amounts of Ammonia can kill almost all the plants in your aquarium. It is harmful to your plants as well as your fish and other living organisms. Therefore, you should monitor ammonium levels in your aquarium periodically to assure the health of the plants and other living organisms. You can test ammonia levels simply, by dipping a test strip into the water. Most chemical strips will simultaneously test ammonia (NH3) and ammonium ions (NH4+).
Ammonia is toxic to plant root systems. It causes extensive damage when allowed to build up quickly. Therefore, it is advised not to use ammonia-contained products such as aquarium fertilizers, especially for new aquariums. Further, the excess ammonia harms aquatic plants due to its high oxidizing ability. It oxidizes highly stable molecules and breaks down organic compounds within aquarium plants and fish, causing toxicity.
Ammonia breaks down the essential compounds which plants require in order to survive and results in the rapid death of plants. Plants may take a shorter time to feel the effects of ammonia toxicity but eventually, the damage will become apparent. Despite the added fertilizers to the aquarium, ammonia is produced by many other processes. Fish poop is the main source of ammonia in an aquarium. Ammonia is a byproduct of fish poop and contains other compounds too.
Many aquarists keep their ammonia level around 1ppm or less, especially in freshwater planted tanks. Some people use an activated carbon filter with a low flow rate and only add water twice a month. In summary, ammonia can be harmful to aquatic life due to its ability to act as a highly corrosive agent killing fragile tissues in living organisms over time.
What is ammonia?
Ammonia is a compound of nitrogen and hydrogen with the formula NH3. It is a naturally formed colorless gas with a distinct pungent smell. This is the building-block chemical and a key component in the manufacture of many products people use every day. It occurs naturally throughout the environment in the air, soil, and water and in plants and animals, including humans. The human body makes ammonia when the body breaks down foods containing protein into amino acids and ammonia, then converting the ammonia into urea.
billions of people around the world use About 90 % of the ammonia to help sustain food production. Ammonia has other important uses including its use in household cleaning products and in manufacturing other products. Although excess amounts cause problems in the aquaculture trade, ammonia is vital to sustaining many important biological processes, such as the nitrogen cycle.

What is the Nitrogen cycle?
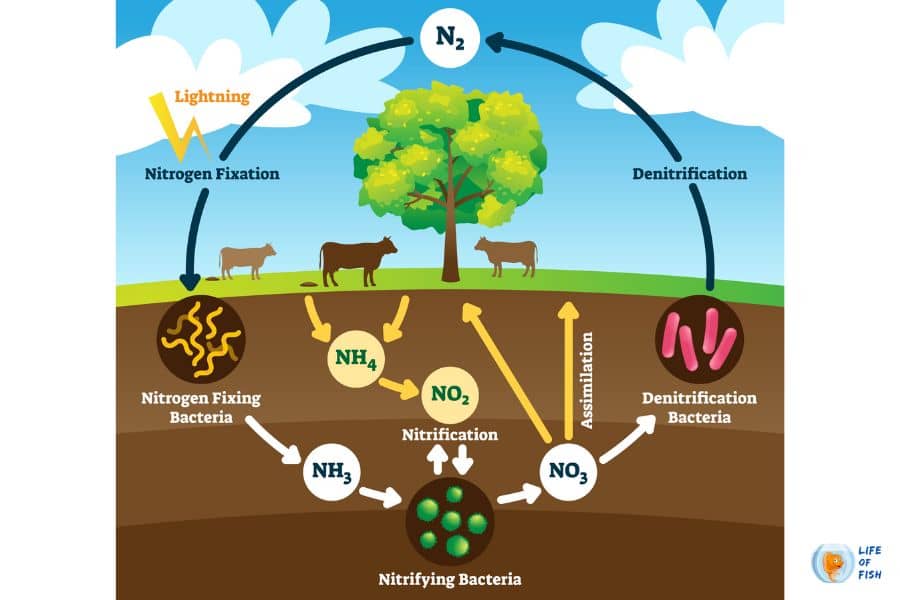
The nitrogen cycle is the cycle through which nitrogen flows in nature in various forms. On a small scale, it can be seen as nitrogen being taken up by plants or animals and then released back when they decompose (through bacteria). Nitrogen, a component of proteins and nucleic acids, is essential to life on Earth. Although 78% of the volume of the atmosphere is nitrogen gas, this abundant reservoir exists in a form unusable by most organisms.
Nitrogen cycles make the nitrogen available to plants, which in turn ultimately sustain all animal life through a series of microbial transformations. The major steps of this process fall into the following classifications: Nitrogen fixation, nitrogen assimilation, ammonification, nitrification, and denitrification.
Nitrogen fixation :
During the process of nitrogen fixation, nitrogen gas is converted into inorganic nitrogen compounds and is mostly (90 percent) accomplished by certain bacteria and blue-green algae. Despite this, a smaller amount of free nitrogen is fixed by abiotic means such as lightning and ultraviolet radiation.
Nitrogen assimilation:
Assimilation is how plants and animals incorporate the NO3- and ammonia formed through nitrogen fixation and nitrification. Plants take up these forms of nitrogen through their roots and incorporate them into plant proteins and nucleic acids. Animals are then able to utilize nitrogen from the plant tissues.
Ammonification:
Assimilation produces large quantities of organic nitrogen, including proteins, amino acids, and nucleic acids. Ammonification is the conversion of organic nitrogen into ammonia. The ammonia produced by this process is excreted into the environment and is then available for either nitrification or assimilation.
Nitrification:
Nitrification is a two-step process in which NH3/ NH4+ is converted to NO3-. First, the soil bacteria Nitrosomonas and Nitrococcus convert NH3 to NO2-, and then another soil bacterium, Nitrobacter, oxidizes NO2- to NO3-. These bacteria gain energy through these conversions, both of which require oxygen to occur.
Denitrification:
Denitrification is the reduction of NO3- to gaseous N2 by anaerobic bacteria. This process only occurs where there is little to no oxygen, such as deep in the soil near the water table. Hence, areas such as wetlands provide a valuable place for reducing excess nitrogen levels via denitrification processes.
Importance of nitrogen in the plant
Nitrogen is among the three macronutrients that are essential for plant function. Further, it is a core component of many plant structures and for both their internal and external metabolic processes.
The wellness of plant parts (leaves, roots, trunks e.t.c) depends on the availability of essential nutrients like nitrogen to enhance the plant’s biological processes including growth, absorption, transportation, and excretion. Since nitrogen is present in different fertilizers, the plants through the roots can enhance uptake.
The functions of nitrogen in plants
- Nitrogen, in a way, could be termed “a backbone” of plants based on what it does in plants. It is required by plants in large amounts since it plays important functions and can be the limiting factor in plant production and proper crop development.
- Nitrogen is an essential element of all the amino acids in plant structures which are the building blocks of plant proteins, important in the growth and development of vital plant tissues and cells like the cell membranes and chlorophyll.
- Nitrogen is a component of nucleic acid that forms DNA, a genetic material significant in the transfer of certain crop traits and characteristics that aid in plant survival. It also helps hold the genetic code in the plant nucleus.
- It is also a component of the chlorophyll molecule. Chlorophyll is important for a plant to capture sunlight energy by photosynthesis which drives plant growth and grain yield.
- Nitrogen is essential in plant processes such as photosynthesis. Thus, plants with sufficient nitrogen will experience high rates of photosynthesis and typically exhibit vigorous plant growth and development.
- This crucial nutrient also plays a role within the plant to ensure energy is available when and where the plant needs it to optimize yield. It helps to regulate water and nutrient intake even present in the plant roots as proteins.
The deficiency of Nitrogen leads to severe plant disorders. It will automatically translate to the reduction of chlorophyll content of plants, therefore, affecting flowering, fruiting, starch, and protein contents undermining plant health.
Will high nitrites kill plants?
Aquarium plants will absorb nitrates from the water, either through their leaves or roots, and use it as a nutrient. Plants can use nitrites as a nitrogen source. High nitrite levels in an aquarium do not kill plants and they will be fine.

Do aquarium plants remove ammonia?
Yes, Aquarium plants have the ability to absorb ammonia from the water of your aquarium. All the live aquarium plants can absorb ammonia in small or large quantities this is very important for the health of the other inhabitants in your tank as Ammonia is toxic for almost all fish species. These plants absorb ammonia and help to improve the water quality of the aquarium. However, the efficiency of absorbing ammonia ranges from 10 to 50%, depending on plant species, growth conditions and the amount of ammonia in water.
Although ammonia is toxic to inhabitants in your aquarium like fish, it is beneficial to your aquarium’s live plants. Ammonia provides nitrogen to the plants. Nitrogen is very important for plant growth as it is essential for the process of photosynthesis. Although all the live aquarium plants can absorb ammonia, faster-growing aquarium plants are good at absorbing ammonia from the tank because they actually require more ammonia to keep up with the growth compared to slow-growing aquarium plants.
Therefore, if you need to remove the ammonia from your tank faster, put fast-growing plants in your tank. The best aquarium plants to remove ammonia from your tank are Pothos, Amazon Sword, Hornwort, Amazon Frogbit, and Java Moss. Besides, ammonia plants also can absorb other nitrogen compounds present in your tanks such as nitrates and nitrites. However, plants prefer ammonia over nitrate and nitrites. This is because, no matter what the nitrogen compound they absorb, first they should convert it to the form of ammonia before using it.

How much ammonia is toxic to aquatic plants?
As mentioned before, excess ammonia can kill your aquarium plants. Therefore, it’s critical to monitor the ammonia levels in your tank. Actually, 1 ppm of ammonia is enough to kill most plants, but it is less toxic at these levels if it is under a PH of 7. Also, the temperature is affected by the extent of the damage. In general, the only safe ammonia level is 0. In conclusion, ammonia levels lower than 1ppm with a pH level lower than 7 and a low temperature is safe for the plants.
Final remarks
Now you know the answer to the question “Will ammonia kill aquarium plants?”. The answer is yes. As the article mentioned excess ammonia is very harmful and deadly for plants. So you should always maintain the ammonium level of your aquarium
Read Next : Aquarium Plants Melting | 4 Proven Reasons |
